How to Benefit from the New Medical Device Ecosystem
The New Medical Device Ecosystem:
A transformation is underway in the realm of medical devices and medical sensor platforms — changing traditional definitions and creating new opportunities.
In this report on Sensor Platforms, Cloud Connectivity, and the New Medical Device Ecosystem, we’ll define what constitutes an ecosystem and how medical device manufacturers can capitalize on collaborative partnerships to achieve a successful medical device in today’s IoMT world. We’ll also explore the transformation of medical devices and medical sensor platforms that’s changing traditional definitions and creating new opportunities; the role of sensor platforms and hardware appliances in expanding medical device functionality and we’ll cover cloud connectivity and keys to a successful medical device ecosystem.
In the new, connected world of the Internet of Medical Things (IoMT), a growing number of medical devices no longer function simply as stand-alone hardware appliances
Think of today’s medical device as an ecosystem formed by a secure, connected, and compliant infrastructure of hardware, sensors, software, user interfaces, and connectivity services – with different types of data and information flowing among these components to the ultimate benefit of the patient.
Fueling this transformation are rapid advances in sensor technology along with the growing use of medical implants, the emerging popularity of smart wearables and mobile devices, and the wider adoption of remote, real-time patient and hardware performance monitoring via cloud-connected platforms.
Challenges and Opportunities
Connected medical devices present opportunities for established players and new entrants especially those who can channel their potential and grasp the reality that competitive pricing and leading quality alone don’t guarantee success in the IoMT world.
By 2026, this new generation of medical devices will drive a global market that is expected to reach USD 176.8 Billion.
With new opportunities come fresh challenges. Unlike several other industries, medical device sensors used in wearable or implantable require design controls and performance metrics to ensure patient safety. Makers of today’s implantable and wearable devices must adhere to strict FDA regulatory guidelines and possess expertise in cloud connectivity, which is often outside their core competency.
This Report
In this report, Galen Data, provider of the industry-leading FDA-compliant Galen Cloud™ platform for connected medical devices, explores the medical device ecosystem in detail and provides guidance on what it takes for manufacturers to build cost effective, compliant, connected medical device solutions.
To unlock value in this next wave of market growth, medical device companies have two alternatives. They can proceed independently by allocating internal resources and acquiring the requisite expertise to launch and maintain a customized cloud-connected solution.
Or, as Galen Data advocates, manufacturers can adopt a collaborative approach and partner with firms who have medical device expertise. This approach can save R&D costs in developing and maintaining connectivity services outside their core competencies.
External experts, with existing robust, turnkey solutions, can provide sensor platforms and cloud connectivity based on industry standards and regulatory requirements, resulting in a medical device ecosystem- based solution. Protecting the intellectual property of the medical device company is also maintained.
As the research firm Deloitte concludes, for medtech companies and device makers to remain competitive, they will need to access talent through “collaborations and partnerships with a diverse range of existing and emerging players, especially academia, engineering companies, data first tech companies and innovative new start-ups.”
We’ll highlight the constituent parts that form the connected medical device ecosystem and how they work together. Then we’ll offer key considerations for success.
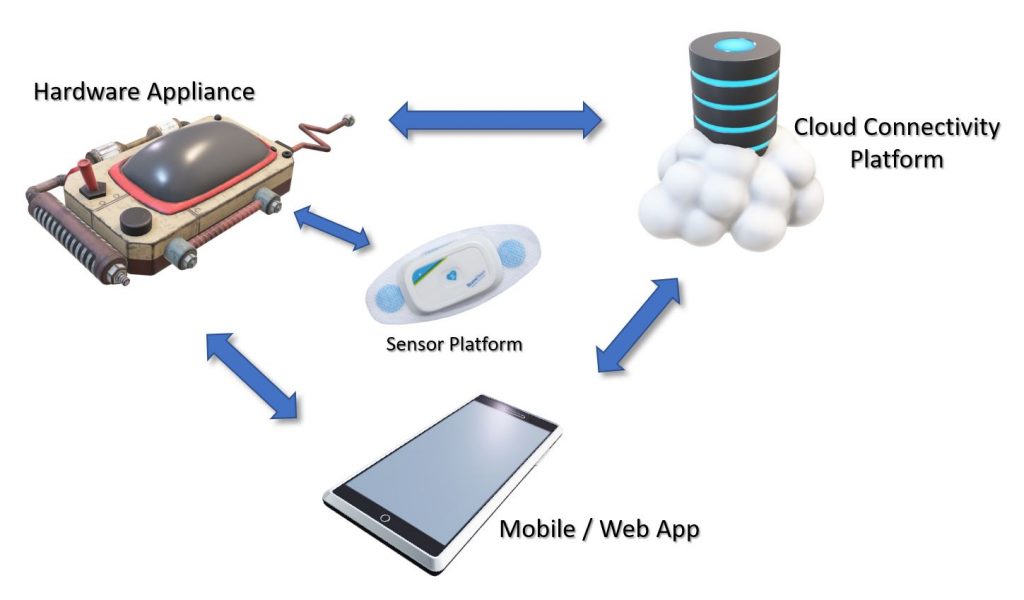
New Medical Device Ecosystem Constituent Parts
To understand the medical device as one ecosystem, it’s first helpful to break it down into constituent parts: the sensor platform (measures patient vital signs), the hardware appliance (applies therapies, communicate wirelessly, displays data), the mobile device that receives the data, and the cloud connectivity platform where the data is collected, stored, shared, and analyzed.
The Hardware Appliance (implantables, wearables, etc.)
Central to the new medical device ecosystem is the instrument and the data it collects.
Examples of medical device ecosystems that can leverage cloud connectivity include:
- Pacemakers
- Defibrillators
- High-frequency ventilators
- Cochlear implants
- Fetal blood sampling monitors
- Neurostimulators
- Insulin pumps
- Medical imaging devices
- And a wide range of wearable vital sign monitors (temperature, heart rate, etc.)
The data generated and collected by devices can include:
- Physiological – temperature, heart rate, vital signs, etc.
- Therapeutic – neurostimulation, drug delivery, etc.
- Demographic – age, sex, location, etc.,
- Device data – battery level, on-board diagnostics, serial number, etc.
Medical devices and the data they collect are carefully regulated by the Food and Drug Administration (FDA) and must go through a rigorous process to test for device safety and effectiveness Under Section 510(k) of the Food, Drug and Cosmetic Act, device manufacturers must register to notify FDA of their intent to market a medical device at least 90 days in advance. This is known as Premarket Notification (PMN).
The FDA assigns devices into one of three classes. Device classification depends on the intended use of the device as well as its indications for use. Classification is also determined by the risks and the level of control needed to ensure a device’s safety and effectiveness. Class I devices generally pose the lowest risk to the patient and/or user and Class III devices the highest risk.
The FDA defines Class III devices as products that “usually sustain or support life, are implanted or present a potential unreasonable risk of illness or injury.” It generally applies to permanent implants, smart medical devices and life support systems.
Devices must demonstrate that measures are in place to guard against vulnerabilities, counter cybersecurity threats, address service reliability, and ensure data is protected if disaster recovery is needed.
Sensor Platform
The sensor, along with sensor platform, is how a medical device initiates patient data.The sensor measures characteristics in its environment, often with electronics whose physical properties change in response to temperature, impedance, capacitance, position/acerbation (motion), and other basic, physical measures. The input could be light, sound, heat, motion, moisture, pressure, or magnetism. The sensor provides as output a weak electrical analog signal, which is amplified, cleaned, and typically converted to a digital signal. That signal is finally converted to a readable display or sent for additional processing where it can then be collected, stored, shared and ultimately analyzed.
The sensor embedded within the hardware appliance generates a signal that is then used in helping a device diagnose, measure, monitor or treat conditions and diseases — ranging from temperature sensors and glucose meters that measure approximate concentration of glucose in the blood, to ECG, and also EMG sensors that track electrical and muscle activity in the heart.
Given their specialized functionality, medical device sensors in wearable or implantable devices need to:
- Comply IEC 60601-1 safety and essential performance standards published by the
International Electrotechnical Commission . Most, if not all, medical device companies
have to comply this widely recognized industry standard that assesses device and
software safety and performance degradation. - Comply with regulatory specifications and standards for “quality management, risk management, usability and functional safety in order to ensure device correctly functions correctly with response to given inputs”
- Deliver precise accurate measurement
- Measure quickly with high stability and fast response time
- Provide digital outputs such as I2C protocols for microcontrollers and microprocessors connectivity
* Source: RF Wireless World
The sensor, along with typically a microprocessor, firmware, the radio that transmits the wireless signal and an interface between the microprocessor and the radio form a sensor platform.
Sensor platforms typically focus on a narrow range of CPT codes for a particular medical surgical, and diagnostic procedure or service.
The sensor platform may be connected using the Bluetooth Low-Energy (BLE) protocol, which is specific for applications where small amounts of data are transmitted at infrequent intervals. BLE is slower and less secure than Wi-Fi but it consumes less energy and therefore is better suited for battery-powered devices.
The signal containing the device’s serial number, patient identification and the data itself may be transmitted and received by a mobile device or base station or docking station.
One such sensor platform is produced by BraveHeart Wireless. Braveheart’s 510(k)-cleared wearable biosensor patches are a part of the BraveHeart platform that enables continuous real time remote monitoring of physiological patient data in a variety of clinical settings to address multiple patient use cases that are reimbursed by a variety of CPT codes.
According to Ted McAleer, vice president of business development at BraveHeart Wireless, a sensor platform should optimally be an open, customizable architecture that provides:
- Configuration for three types of data delivery to manage efficiency: pure streaming, store
and forward, and snapshot modes. Pure streaming is data that is continuously generated.
Store and forward is a process where data is transmitted and temporarily stored before
being forwarded to the destination node. Snapshot mode as the name implies captures data
a specific point in time; it’s useful for data backup while consuming less storage space. - Modular hardware architecture (to enable multiple sensor packages for different client and use cases)
- Open RESTful API for easy integration and interoperability.
- Superior Bluetooth Low-Energy performance
- Battery management/performance
- The ready ability to Integrate into the electronic health record (EHR) and other monitoring applications and dashboards
Cloud Platforms
In addition to sensor platforms and hardware appliances, the third component in the connected device ecosystem is the cloud platform.
Mobile devices, base station or docking station house a cloud platform’s API. An API (Application Programming Interface) is proprietary software that allows two applications to talk to each other.
Through a secure Wi-Fi or cell connection, encrypted data can be sent to the cloud where it can be collected, stored, shared, accessed and analyzed. In the Galen Cloud, data display can be customized and workflows for data easily created to allow for post processing. Manufacturers, providers, and patients can access this data and receive defined push notifications.
As Galen Data CEO Chris DuPont likes to point out, “Unlike automotive dashboards, medical devices don’t have a check engine light that alerts when the device is not working properly.”
The ability to leverage the cloud to collect and analyze diagnostic data characterizing the performance and status of the device provides that check engine light.
The cloud platform allows manufacturers to monitor devices remotely and transmit software updates directly to the device without medical intervention by a provider — which is considerably more convenient, safer, and less expensive to perform.
Visit Benefits of Cloud Connectivity
Keys to a Successful Medical Device Ecosystem
Collaborative, Turnkey Solutions
As stated in the outset, cloud-connected medical devices pose challenges and opportunities. For those who lack the resources, internal expertise, and regulatory knowledge, their best option may be to collaborate with experienced partners who can help develop, launch, and scale their solution.
Partnering requires a different mindset for engineers who view solving problems on their own as a source of great pride. (As engineers ourselves, we can appreciate your mindset.)
But increasingly, traditional medical device companies who recognize the value of cloud connectivity are turning to companies like Galen Data and sensor platform manufacturer BraveHeart to optimize resources. Their turnkey solutions minimize software customization. They enable firms to reduce design costs and avoid delays to important product lifecycle milestones such as clinical trials, FDA clearance, and eventual market implementation.

BraveHeart Bravo1 Patch sensor platform
As BraveHeart Wireless’ vice president of business development Ted McAleer states, “Historical software platforms that have resided in traditional medical devices are not ‘purpose built’ for data sharing; In an IoMT world, that’s no longer a viable option. Medical device manufacturers need sensor platform designers who are equipped to handle the demands of a cloud-connected environment and data interoperability.”
Quality Systems and Security and Privacy Protocols
Collaboration starts with nimble, quality-centric partners who understand the hardware and software architecture associated with cloud connectivity.
Look for firms who adhere to quality systems and best-in-class protocols for security and privacy and safeguard against vulnerabilities that can compromise data integrity and undermine patient health. Regulatory requirements and industry standards for medical device connectivity can be overwhelming and the classification guidelines confusing for companies unfamiliar with the process.
The Galen Cloud, for example, certified under ISO 13485:2016 (Medical Devices – Quality Management Systems). Additionally, the Galen Cloud:
- Is compliant to FDA, European MDR, and Health Canada regulations.
- Adheres to HIPAA, EU General Data Protection Regulations (GDPR), and the California Consumer Privacy Act (CCPA).
- Is developed per FDA Design Controls (21 CFR Part 820), IEC 62304 (Medical Device Software – Software Lifecycle Processes), and ISO 14971 (Application of Risk Management to Medical Devices).
Data-Focused
The medical device ecosystem requires manufacturers to see their business in a whole new light.
Their market potential is far more than the patient-focused functionality of their instruments alone; it also exists in the data their devices generate. And its currency will only increase in a value-based system of care driven by analytics and software-based services and solutions.
As we discussed in Opportunities and Pitfalls for Connected Medical Devices, “Because data is collected centrally in the cloud, engineers and researchers can improve diagnostic algorithms over time. Machine learning techniques can be used to comb through this data and identify new patterns that can lead to new products or services. Connected medical devices can also provide portable diagnostics devices that can be used for in-home collection and diagnosis.”
The connected medical device ecosystem makes data’s impact possible.
Conclusion – Benefits of the New Medical Device Ecosystem
Manufacturers will benefit from an exciting new era where their medical device technology is now an ecosystem of hardware, sensor platforms, web applications, and cloud connectivity.
Today’s connected medical devices will open lucrative markets for new and existing companies including remote monitoring, data analytics, and machine learning. It will allow devices to interoperate with EHR systems to track patient health and wellness.
Success will require new ways of viewing medical devices themselves – what they can do and how they are built. Given devices’ growing complexity, companies will need to embrace an approach that is:
- Collaborative – based on partnerships that take advantage of outside expertise in cloud connectivity, regulatory compliance issues, and quality control measures.
- Data Focused — understanding the value of a device is increasingly derived from the data it generates.
The most successful collaborations result when partners can combine the incentive for profit with a shared vision of innovation and a commitment to improving patient health, reducing the cost of care, and providing easier access.