Where is Digital Health Heading in a Post-Covid World?
When the COVID-19 pandemic hit in early 2020, there was an almost immediate uptake in digital healthcare solutions. The massive digital transformation that took place out of necessity in a short time-frame showed the healthcare industry that new approaches were entirely possible.
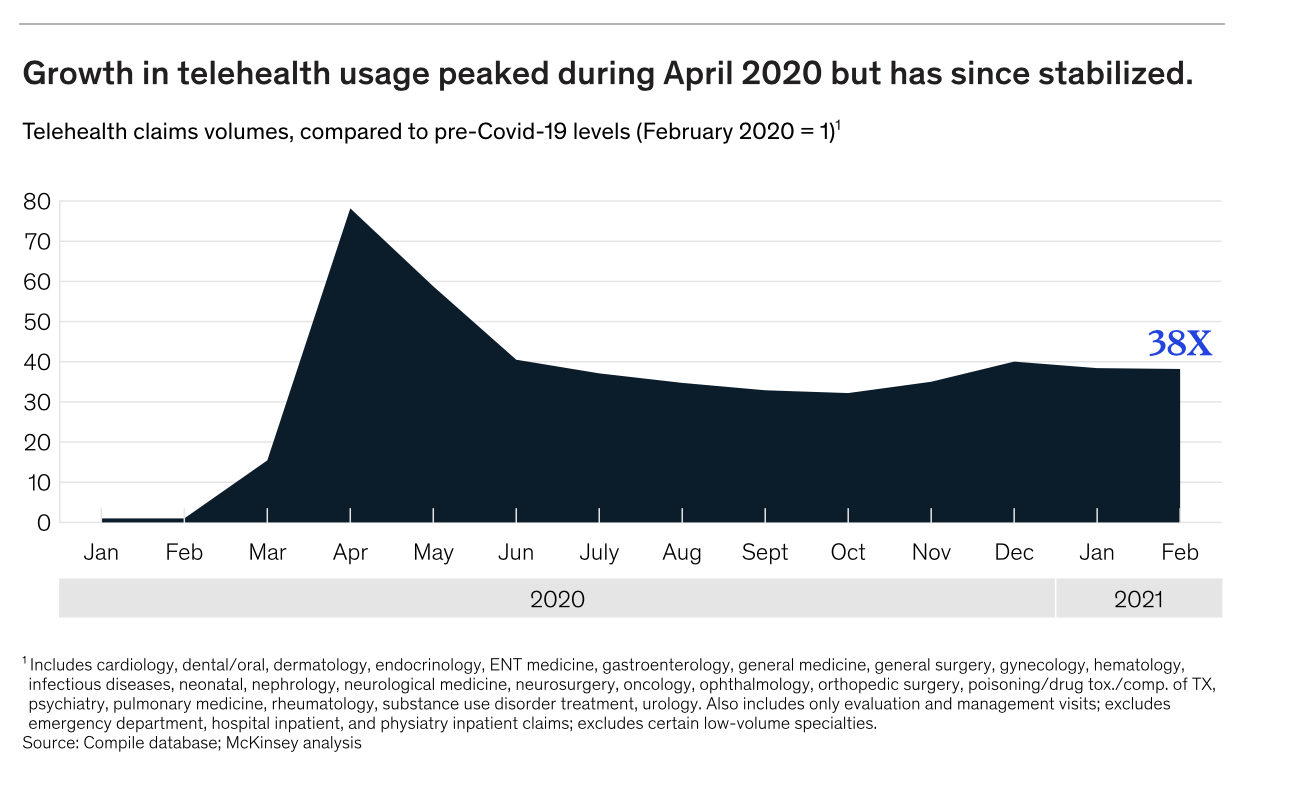
A McKinsey and Company analysis found that growth in telehealth peaked in April of 2020. While that growth has since stabilized, it still sits at significantly higher adoption rates than in pre-pandemic times.
While the switch to digital health solutions was borne out of necessity, this did allow a period of time for digital technologies to “prove themselves”, both to physicians and their patients. In a post-Covid world, digital healthcare offers a reinvention of traditional models, as it offers benefits such as access, affordability, and positive patient health outcomes.
Where is digital health heading? Here’s what we think:
Improved physician and patient perception
One of the challenges for digital health solutions has always been the perceptions of physicians and patients. Slow adoption has often been due to data security concerns, worries about accuracy or efficacy, and a perception that a digital health technology solution just “won’t be as good” as an in-person consult.
In effect, the pandemic forced the issue, with many having no choice but to adopt innovative technology solutions to continue care at a distance. For example, digital health tracking tools saw their biggest growth in adoption, rising from 42% in 2019 to 54% adoption in 2020. In effect, these digital health technology tools were given the chance to prove themselves where perhaps they may not otherwise have been adopted so quickly.
With more physicians and patients exposed to digital health solutions and seeing how well they can work, they’ll be more inclined to work with digital health companies to adopt new innovative tools. It’s clear, the perception of digital technologies being able to be used in healthcare is improving. This is especially important as emerging technologies, such as artificial intelligence, continue to mature. In a post-covid world, it’s more likely professionals in the healthcare industry will be more willing to try a digital solution. For developers of these solutions, this is a positive step toward growth in adoption, as well as the ability to get approval for programs such as Medicare.
McKinsey and Company found that overall, attitudes toward telehealth solutions have improved, although there is still room to grow. “Some barriers—such as perceptions of technology security—remain to be addressed to sustain consumer and provider virtual health adoption, and models are likely to evolve to optimize hybrid virtual and in-person care delivery.”
Telehealth is here to stay
Adoption of telehealth prior to Covid-19 was relatively small. During the pandemic, physicians saw telehealth visit volumes soar by a factor of 50 to 175. In a post-covid world, 40% of consumers believe they will continue to use telehealth in the future, up from 11% pre-pandemic. There is no doubt, the future of healthcare will include telehealth.
Additionally, some key regulatory changes that granted telehealth as a solution during the pandemic have been made permanent. This means reimbursements for telehealth will continue to be available.
In other words, telehealth is here to stay. While many providers had to hastily construct solutions to bridge the gap early in the pandemic, a more permanent presence of telehealth means that healthcare systems need to ensure they have robust digital solutions, including approaches to help patients, not just for initial medical evaluations, but to treat chronic conditions as well.
Telehealth technology needs to integrate with electronic health records and clinical protocols for it to work on a more permanent basis. Additional questions remain, for example, when is telehealth appropriate or not appropriate? How will healthcare organizations ensure the patient experience is positive? How will they monitor that the outcomes of telehealth visits compare favorably to office visits?
Delivering care in the right setting will be a key focus. Telehealth has the potential to relieve ER visits and support chronic care management – it’s a matter of having the protocols in place.
Investors want digital health solutions
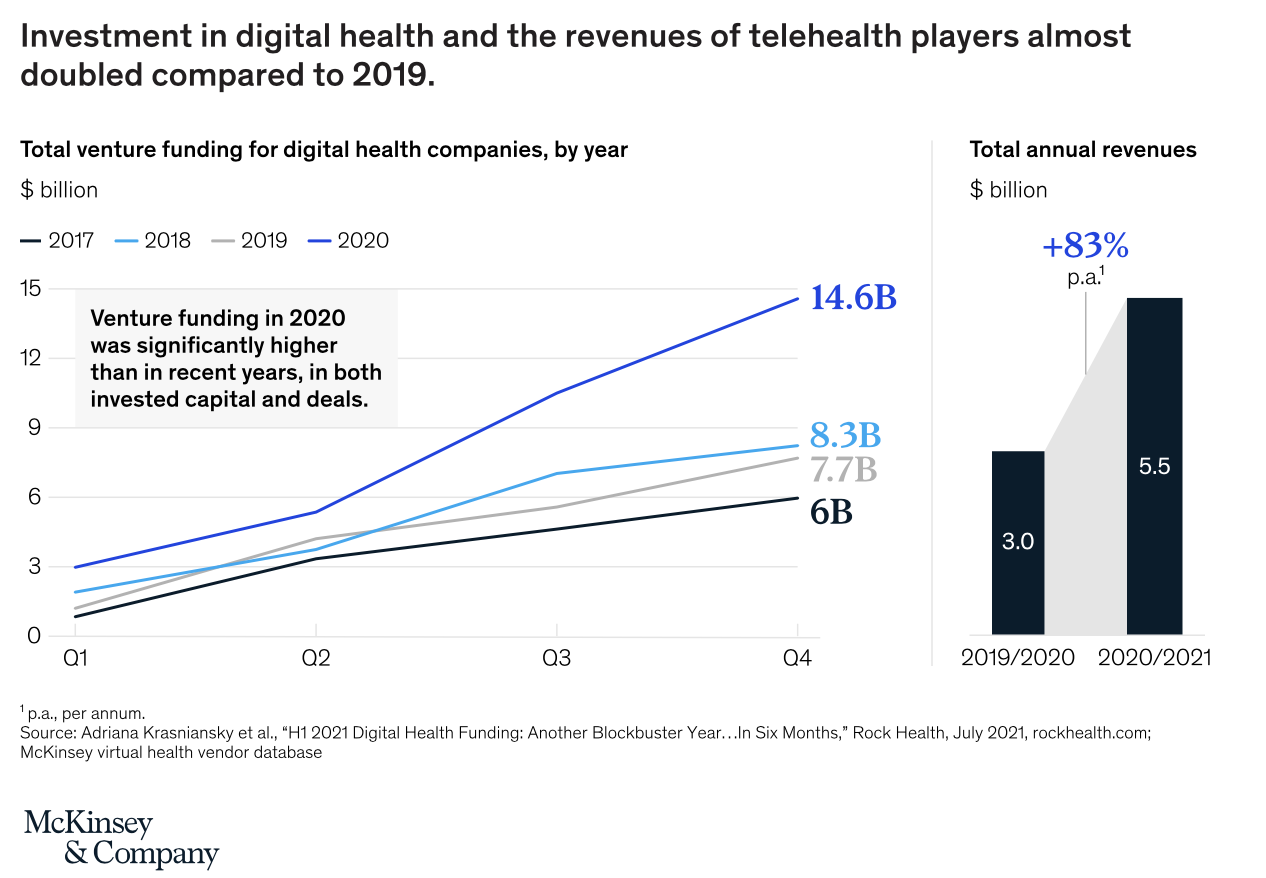
Importantly for developers of medical devices and digital health solutions, investor interest is accelerating. From McKinsey and Company: “Per Rock Health’s H1 2021 digital health funding report the total venture capital investment into the digital health space in the first half of 2021 totaled $14.7 billion, which is more than all of the investment in 2020 ($14.6 billion) and nearly twice the investment in 2019 ($7.7 billion).”
For medical device companies needing to attract funds, this is good news for the future of digital health development. Investors are looking for innovation in this space and there is plenty of room for companies to take up this mantle. Those with high-quality products will find they have more potential options for funding than they ever did previously.
Improved data flow is in demand
One of the ongoing challenges for those in the digital health space is that there is demand for improved data flow or integration across systems. For example, if a patient is wearing a monitoring device that gathers information on blood pressure, how will that data safely and securely be added to their patient record held on a different software system?
Effective hybrid care systems, where clinicians see patients both in-person and virtually, rely on high-quality data transfer to inform them. One thing device manufacturers can do is build device connectivity with data integration in mind. Physicians and patients can be overwhelmed by multiple independent systems, but uptake is likely to be higher when that challenge is overcome.
Connected medical devices drive innovation
It’s fair to say that the pandemic sparked more innovation in connected medical devices. This development is set to continue in a post-covid world, particularly in light of patient, physician, and investor demand.
One thing that has been highlighted across the pandemic has been the strain on healthcare workers. This shines a light on potential digital solutions to help ease that burden, including connected devices that allow rapid sharing of key data and the possibility of decreased response times.
The pandemic has also contributed to key knowledge about the challenges associated with healthcare when “at a distance” is preferred. For example, there are innovations happening with medical devices that haven’t traditionally been connected. Ventilators are one device among these.
Another growth area among connected devices is the use of biosensors. These sensors, which can be worn, ingested, or optical, have applications for screening, early detection, health management, surveillance, and even chronic disease treatment. Biosensors are scalable and offer the potential to lower costs for patients and providers. Data collection can be linked to real-time decision-making, with the potential to improve patient health outcomes.
Emphasis on security and reliability
Cybersecurity and the assurance that the information gathered by digital health solutions is reliable is the subject of ongoing scrutiny and increased emphasis. It is expected that we’ll see more amendments to regulations in the coming years, especially as artificial intelligence is more fully integrated.
Currently, manufacturers of connected devices should look to security standards from IEC 62304, risk management standards from ISO 14971, FDA guidance documents on cybersecurity, and if entering the EU, MDR, IVDR, the guidance document MDCG 2019-16.
It’s important to build your device on a secure platform that meets those regulations, which can be an intensive operation. An alternative to building it yourself from the ground up is to use a cloud platform that has been specially built for medical devices. This ensures that you get a compliant solution from the beginning, helping innovative solutions to get to market more quickly.
Galen Data provides a purpose-built cloud platform for medical devices. Take a look at our solution here and be part of the digital health revolution. The future of healthcare is, without a doubt, going to be more digital.